How a Top 10 Pharma Company Transformed Clinical Program Evaluation with Causaly
How a Top 10 Pharma Company Transformed Clinical Program Evaluation with Causaly
Challenges
A top 10 pharma company tasked its External Innovation Advisor with evaluating external clinical and preclinical programs to inform indication-level strategy. This required systematic benchmarking of assets by mechanism, modality, and development stage to guide partnership decisions and internal prioritization.
The work demanded scientific rigor and speed. Strategic decisions depended on accurate interpretation of clinical outcomes, program discontinuations, and emerging data across competitive therapeutic landscapes.
To inform strategy, the advisor needed clear, defensible answers to critical questions:
- How are comparable programs performing in the clinic?
- Where have similar approaches failed or been discontinued?
- Do observed outcomes reflect biological signal or trial design effects?
The existing process relied on manual searches across PubMed, clinical trial registries, conference abstracts, and secondary literature. The advisor began with keyword searches, moved to review articles, and then chased references to validate claims and reconstruct program histories.
“I used to start with PubMed and clinical databases, find a recent review, and then chase references and abstracts, sometimes for days.”
Even after days of work, the resulting landscape remained fragmented. Evidence was spread across multiple sources and formats. Spreadsheet aggregation introduced risk. New data forced repeated rebuilds. The process slowed decision-making and increased the chance of missing critical evidence.
How Causaly Helped
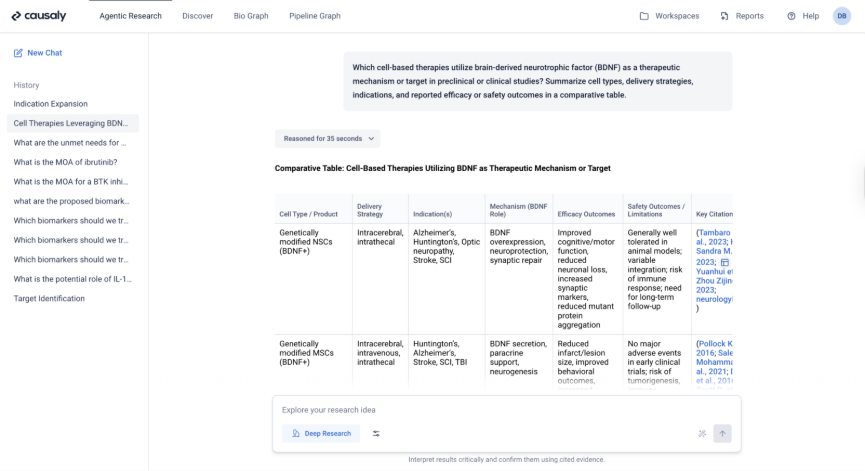
Replace Broad Searches with Structured Evidence Discovery
Instead of starting with open-ended database searches, the advisor used Causaly's Agentic Research to query specific targets, mechanisms, modalities, and indications.
Causaly surfaced:
- Structured relationships extracted from primary literature
- Relevant clinical and preclinical evidence in context
- Direct links to source publications
The advisor built a comprehensive evidence foundation in minutes rather than days.
Benchmark Clinical Outcomes Across Comparable Programs
To assess competitive performance, the advisor used Causaly's Pipeline Graph:
- To query trials by target, modality, and indication
- Compare endpoints, patient populations, and study designs
- Review structured summaries linked directly to trial records
Causaly eliminated manual aggregation and enabled real-time benchmarking. All outputs were evidence-linked and ready for internal review, reducing the need for spreadsheets and ad hoc tracking.
“Now, I can get the same depth in minutes and if it outputs a table, I can pull the exact references behind each point. It’s faster and reduces the risk of missing key perspectives.”
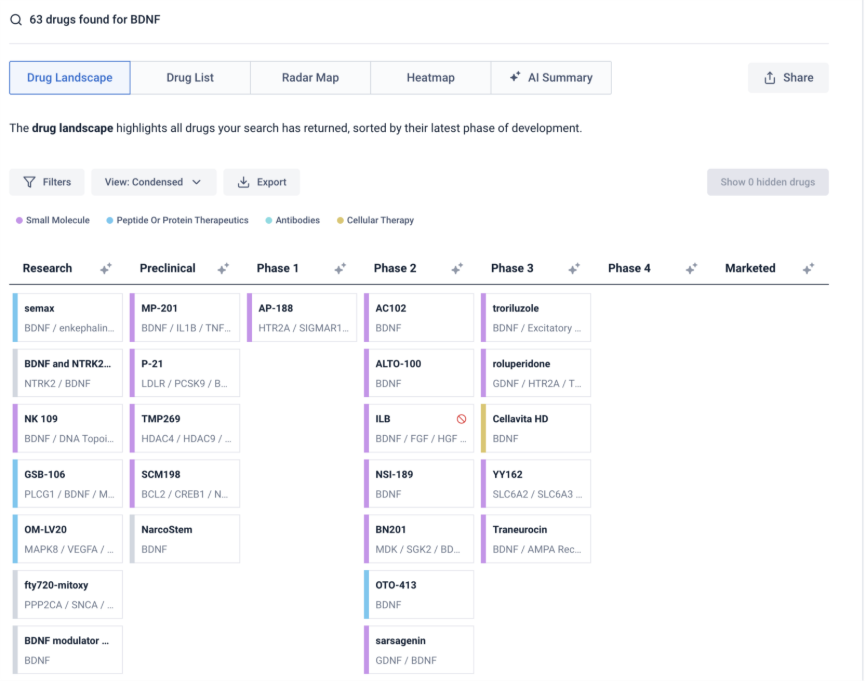

Systematically Analyze Failures and Discontinuations
Rather than relying on anecdotal reports, the advisor:
- Identified discontinuations associated with specific mechanisms
- Compared modality-specific failure patterns
- Reviewed citation-backed evidence behind each outcome
This approach revealed patterns across programs and indications with traceable evidence.
Distinguish Biological Signal from Trial Design Effects
To evaluate whether outcomes reflected real biological activity, the advisor:
- Compared results across multiple studies
- Examined consistency across trial designs
- Assessed endpoint selection and patient stratification
Causaly linked every conclusion to primary sources, strengthening scientific confidence.
Instead of rebuilding analyses from scratch, the advisor refreshed queries as new data emerged. Evidence tables remained citation-linked and export-ready for internal discussion.
The workflow became iterative, scalable, and sustainable.
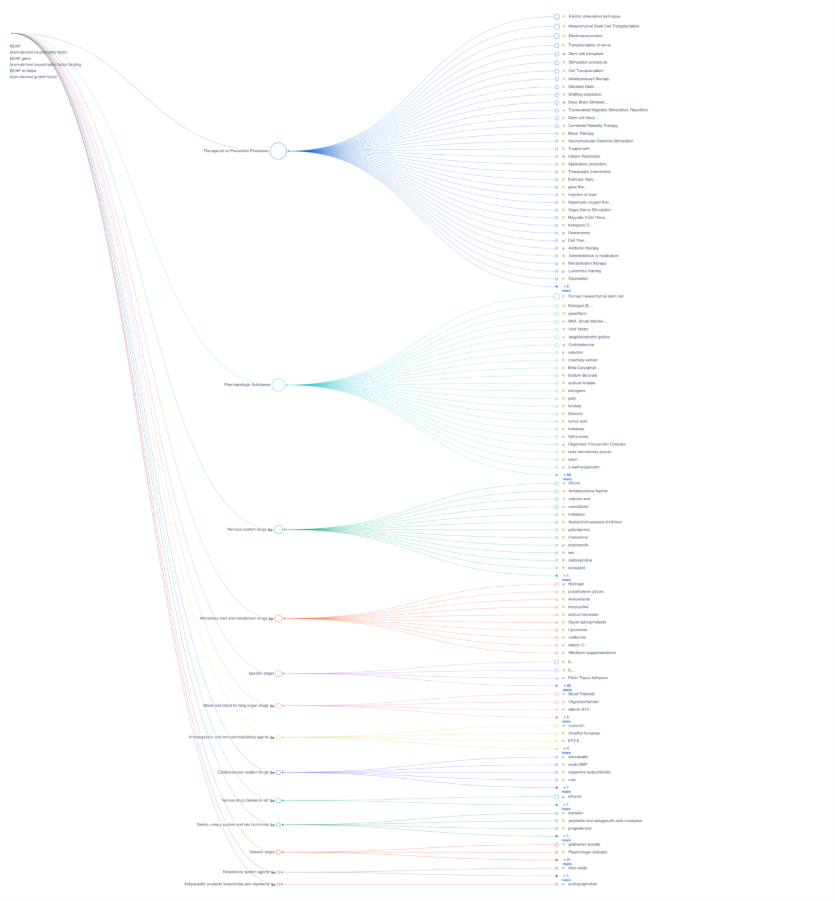

Impact
- The advisor accelerated clinical landscape analysis from multi-day manual reviews to minutes while maintaining full traceability to source evidence.
- Cross-study, citation-backed outputs increased confidence and stronger interpretation of clinical outcomes when evaluating differentiation, biological validity, and development risk.
- Faster strategic alignment with Clear, evidence-linked summaries improved discussions across clinical, translational, and strategy teams, reduced rework, and accelerated decision-making.
I used to start with PubMed and clinical databases, find a recent review, and then chase references and abstracts, sometimes for days. Now, I can get the same depth in minutes and if it outputs a table, I can pull the exact references behind each point. It’s faster and reduces the risk of missing key perspectives.


Get started with Causaly

Ready to transform the way your R&D teams discover and deliver? Take the first step - see Causaly for yourself.
Request a demo

